热带海洋学报 ›› 2022, Vol. 41 ›› Issue (1): 28-41.doi: 10.11978/2021025CSTR: 32234.14.2021025
比较基因组学分析弧菌属群体感应通路的分布与进化
- 1. 中国科学院热带海洋生物资源与生态重点实验室, 广东省海洋药物重点实验室, 中国科学院南海海洋研究所, 广东 广州, 510301
2. 中国科学院大学, 北京 100049
-
收稿日期:2021-02-24修回日期:2021-04-15出版日期:2022-01-10发布日期:2021-04-22 -
通讯作者:高贝乐 -
作者简介:毛颖津(1994—), 男, 广东省中山市人, 硕士研究生, 研究方向为微生物功能基因组学研究。email:maoyingjin18@mails.ucas.edu.cn -
基金资助:广州市科技计划项目(201804010437)
Comparative genomic analysis of the distribution and evolution of quorum sensing pathways in the Vibrio genus
MAO Yingjin1,2( ), GAO Beile1(
), GAO Beile1( )
)
- 1. CAS Key Laboratory of Tropical Marine Bio-resources and Ecology, Guangdong Key Laboratory of Marine Materia Medica, South China Sea Institute of Oceanology, Chinese Academy of Sciences, Guangzhou 510301
2. University of Chinese Academy of Sciences, Beijing 100049
-
Received:2021-02-24Revised:2021-04-15Online:2022-01-10Published:2021-04-22 -
Contact:GAO Beile -
Supported by:Science and Technology Program of Guangzhou, China(201804010437)
摘要:
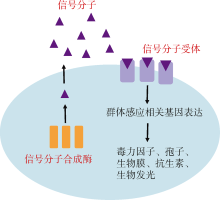
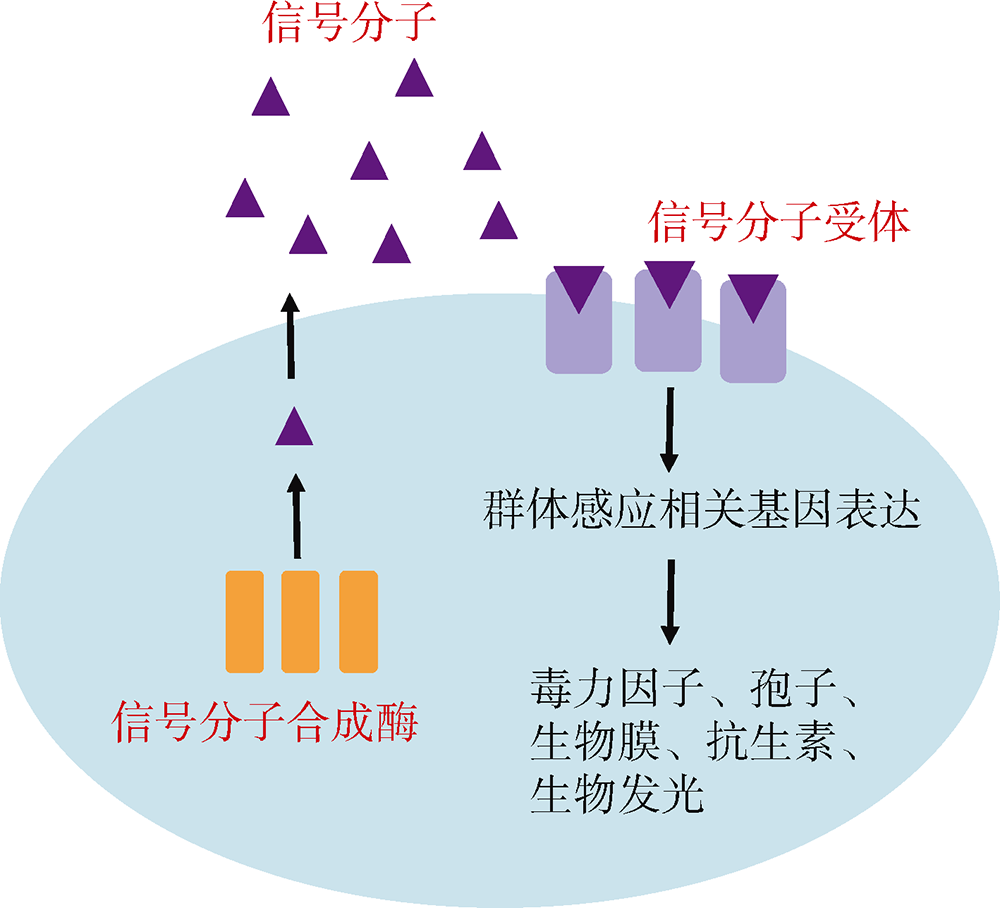
群体感应是一种细菌细胞间的通讯过程, 细菌通过测量细胞外自诱导剂浓度从而感知群体细胞密度变化。群体感应使细菌能够在两种基因表达模式下转换: 在低细胞密度时有利于个体发展, 而在高细胞密度时则有利于群体发展。目前主要有7种群体感应通路, 分别以oligopeptides、AHL(Acylated Homoserine Lactones)、AI-2(Autoinducer-2)、CAI-1(Cholera Autoinducer-1)、PQS(Pseudomonas Quinolone Signal)、AI-3(Autoinducer-3)以及DSF(Diffusible Signal Factor)作为各群体感应通路的信号分子; 其中oligopeptides存在于革兰氏阳性细菌中, 其余6种信号分子存在于革兰氏阴性细菌中。弧菌是导致人类感染和水产品污染的主要病原菌, 传统的抗生素治疗弧菌感染具有很强的选择压力, 导致越来越多耐药弧菌出现。面对愈发严峻的弧菌耐药性问题, 目前群体感应淬灭被认为是治疗弧菌感染的重要替代手段之一, 因而有必要调查弧菌属细菌不同群体感应通路的分布情况, 为针对弧菌的群体感应淬灭剂研发提供重要参考信息。通过比较基因组学分析, 发现全基因组测序的46株弧菌存在AHL、AI-2以及CAI-1这3种信号分子通路, 其中5株弧菌均含有上述3种信号分子通路, 共有30株弧菌只含有AI-2和CAI-1两种信号分子通路; 而46株弧菌都不存在PQS、AI-3以及DSF群体感应通路。此外, 弧菌群体感应通路的分布与物种进化有关, 含有同种群体感应信号分子通路的弧菌, 其亲缘关系较近, 说明该通路的基因进化自它们的共同祖先。本研究表明开发针对弧菌的群体感应淬灭剂应以AI-2和CAI-1两种信号分子介导的通路作为靶点。
中图分类号:
- P735.51
引用本文
毛颖津, 高贝乐. 比较基因组学分析弧菌属群体感应通路的分布与进化[J]. 热带海洋学报, 2022, 41(1): 28-41.
MAO Yingjin, GAO Beile. Comparative genomic analysis of the distribution and evolution of quorum sensing pathways in the Vibrio genus[J]. Journal of Tropical Oceanography, 2022, 41(1): 28-41.
表1
革兰氏阴性菌群体感应通路"
| 合成酶 | 信号分子 | 受体蛋白 | 模式菌株 | 参考文献 |
|---|---|---|---|---|
| LuxI | AHL | LuxR | 费氏弧菌 Aliivibrio fischeri | Eberhard et al, |
| LuxS | AI-2 | LuxPQ/LsrB/RbsB | 霍乱弧菌 Vibrio cholerae | Bassler et al, |
| CqsA | CAI-1 | CqsS | 霍乱弧菌 Vibrio cholerae | Miller et al, |
| PqsA/B/C/D/E/H | PQS | PqsR | 铜绿假单胞菌 Pseudomonas aeruginosa | Pesci et al, |
| LuxS | AI-3 | QseBC/QseEF | 大肠杆菌 Escherichia coli O157:H7 | Sperandio et al, |
| RpfF | DSF | RpfCG/RpfR | 野油菜黄单胞菌 Xanthomonas campestris | Barber et al, |
表2
菌株信息"
| 物种名称 | 菌株 | 基因组编号 | 基因组大小/Mb | 编码序列数目/个 |
|---|---|---|---|---|
| Vibrio alfacsensis | CAIM 1831 | GCA_003544875.1 | 4.91 | 3830 |
| Vibrio alginolyticus | FA2 | GCA_011801435.1 | 5.25 | 4598 |
| Vibrio anguillarum | VIB12 | GCA_002310335.1 | 4.90 | 4091 |
| Vibrio antiquarius | EX25 | GCA_000024825.1 | 5.09 | 4451 |
| Vibrio aphrogenes | CA-1004 | GCA_002157735.2 | 3.38 | 2881 |
| Vibrio astriarenae | HN897 | GCA_010587385.1 | 4.80 | 4176 |
| Vibrio breoganii | FF50 | GCA_001677275.1 | 4.49 | 3978 |
| Vibrio campbellii | BoB-90 | GCA_002906455.1 | 6.17 | 5407 |
| Vibrio casei | DSM 22364 | GCA_002218025.2 | 4.14 | 3601 |
| Vibrio chagasii | ECSMB14107 | GCA_004022545.1 | 5.55 | 4448 |
| 物种名称 | 菌株 | 基因组编号 | 基因组大小/Mb | 编码序列数目/个 |
| Vibrio cholerae | 10432-62 | GCA_000969265.1 | 4.08 | 3546 |
| Vibrio cidicii | 2756-81 | GCA_009763805.1 | 4.75 | 3492 |
| Vibrio cincinnatiensis | 2070-81 | GCA_009763705.1 | 3.81 | 3284 |
| Vibrio coralliilyticus | RE22 | GCA_003391375.1 | 5.78 | 5064 |
| Vibrio cyclitrophicus | ECSMB14105 | GCA_005144905.1 | 5.07 | 4283 |
| Vibrio diabolicus | FA3 | GCA_011801455.1 | 5.11 | 4501 |
| Vibrio europaeus | NPI-1 | GCA_013154935.1 | 5.45 | 4782 |
| Vibrio fluvialis | FDAARGOS_104 | GCA_001558415.2 | 4.83 | 4338 |
| Vibrio furnissii | FDAARGOS_777 | GCA_006364355.1 | 4.99 | 4466 |
| Vibrio gazogenes | ATCC 43942 | GCA_002196515.1 | 4.79 | 4070 |
| Vibrio harveyi | ATCC 33843 | GCA_000770115.2 | 5.88 | 5166 |
| Vibrio hyugaensis | 090810a | GCA_002906655.1 | 5.61 | 4845 |
| Vibrio jasicida | 090810c | GCA_002887615.1 | 5.99 | 5188 |
| Vibrio mediterranei | QT6D1 | GCA_002214345.1 | 5.81 | 5146 |
| Vibrio metoecus | 08-2459 | GCA_009665275.1 | 3.99 | 3518 |
| Vibrio metschnikovii | 9502-00 | GCA_009763765.1 | 3.62 | 3118 |
| Vibrio mimicus | SCCF01 | GCA_001767355.1 | 4.48 | 3925 |
| Vibrio natriegens | CCUG 16371 | GCA_001680045.1 | 5.16 | 4451 |
| Vibrio navarrensis | 2462-79 | GCA_009763725.1 | 4.78 | 4093 |
| Vibrio nigripulchritudo | SFn1 | GCA_000801275.1 | 6.32 | 5545 |
| Vibrio owensii | XSBZ03 | GCA_002021755.1 | 5.89 | 5112 |
| Vibrio panuliri | JCM 19500 | GCA_009938205.1 | 4.86 | 4235 |
| Vibrio ponticus | DSM 16217 | GCA_009938225.1 | 4.80 | 4098 |
| Vibrio parahaemolyticus | VPD14 | GCA_004006515.1 | 5.17 | 4531 |
| Vibrio qinghaiensis | Q67 | GCA_002257545.1 | 4.02 | 3373 |
| Vibrio rotiferianus | B64D1 | GCA_002214395.1 | 5.28 | 4593 |
| Vibrio rumoiensis | FERM P-14531 | GCA_002218045.2 | 4.21 | 3605 |
| Vibrio scophthalmi | VS-12 | GCA_001685465.1 | 4.93 | 4206 |
| Vibrio sp | 2521-89 | GCA_002216685.1 | 4.12 | 3539 |
| Vibrio splendidus | BST398 | GCA_003345295.1 | 5.51 | 4582 |
| Vibrio taketomensis | C4III291 | GCA_009938185.1 | 4.36 | 3526 |
| Vibrio tapetis | CECT4600 | GCA_900233005.1 | 5.73 | 4800 |
| Vibrio tasmaniensis | LGP32 | GCA_000091465.1 | 4.97 | 4169 |
| Vibrio tritonius | JCM 16456 | GCA_001547935.1 | 5.22 | 4549 |
| Vibrio tubiashii | ATCC 19109 | GCA_000772105.1 | 5.54 | 4946 |
| Vibrio vulnificus | 07-2444 | GCA_009764115.1 | 5.23 | 4554 |
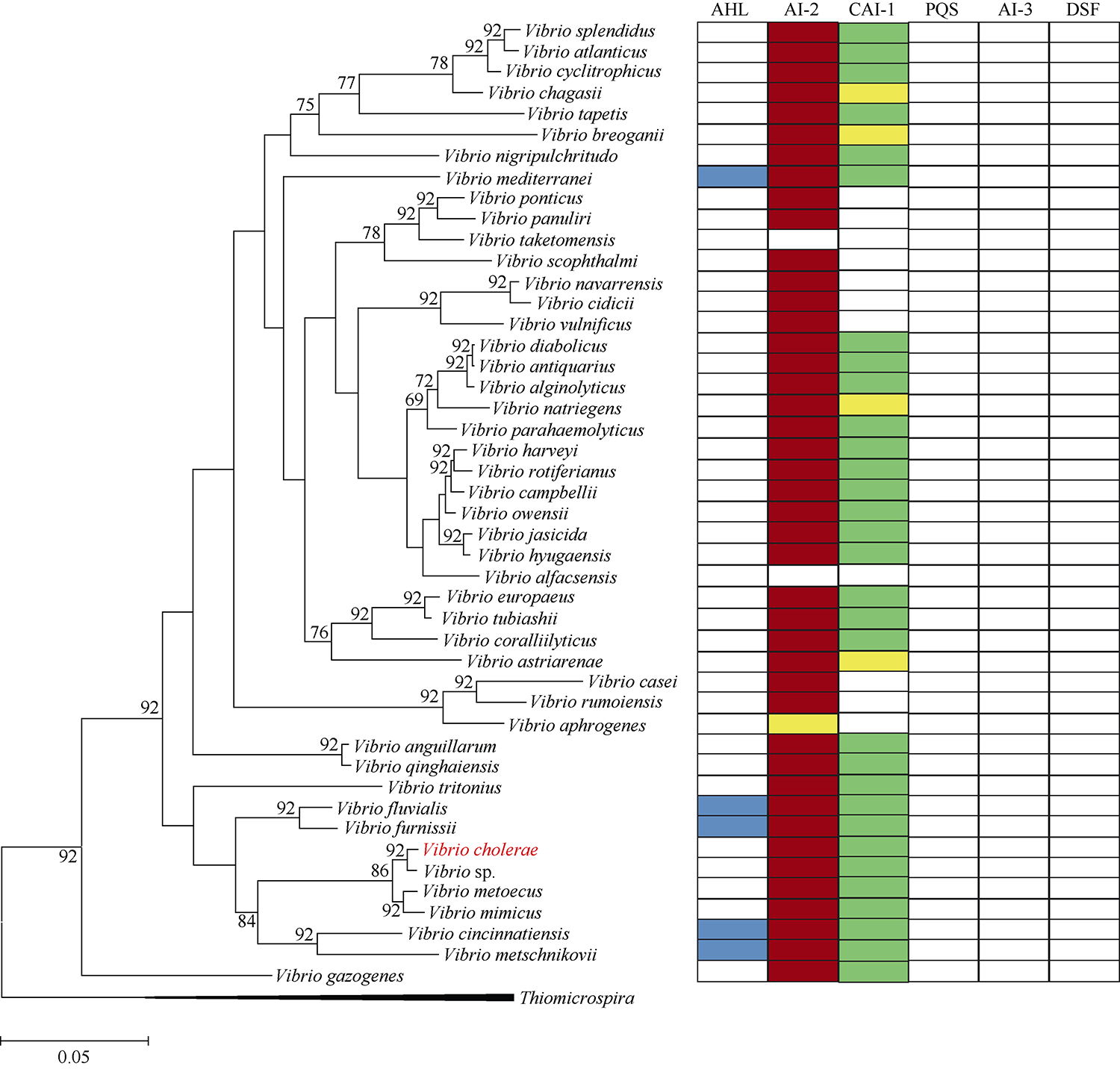
图2
46株弧菌的系统进化树 线段0.05代表1/200进化距离单位; 分支上的数值为自举1000次的结果; 分支上数值的阈值设置为70, 低于70不显示; 左侧进化树上用红色字体标注表明在Vibrio cholerae中分别发现了AI-2和CAI-1两种信号分子通路; 进化树选择目前已全基因组测序的3株Thiomicrospira作为进化树的外枝。右侧表格中蓝色、红色和绿色方格分别表示目的菌株含有对应的AHL、AI-2或CAI-1信号分子通路, 黄色方格代表根据进化树上与目的菌株亲缘关系相近的其他弧菌含有AI-2或CAI-1信号分子通路而推测目的菌株可能也含有相应的信号通路, 但该通路的合成酶和受体编码基因在基因组上没有相邻分布; 白色方格表示不含该种信号通路的菌株"
| [1] | 郑林, 祝令伟, 郭学军, 等, 2019. 副溶血性弧菌耐药基因的研究进展[J]. 中国兽药杂志, 53(6): 80-85. |
| ZHENG LIN, ZHU LINGWEI, GUO XUEJUN, et al, 2019. Advances in antimicrobial resistance genes in Vibrio parahaemolyticus[J]. Chinese Journal of Veterinary Drug, 53(6): 80-85. (in Chinese with English abstract) | |
| [2] |
ALTSCHUL S F, MADDEN T L, SCHÄFFER A A, et al, 1997. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs[J]. Nucleic Acids Research, 25(17): 3389-3402.
doi: 10.1093/nar/25.17.3389 |
| [3] |
BARBER C E, TANG J L, FENG J X, et al, 1997. A novel regulatory system required for pathogenicity of Xanthomonas campestris is mediated by a small diffusible signal molecule[J]. Molecular Microbiology, 24(3): 555-566.
doi: 10.1046/j.1365-2958.1997.3721736.x |
| [4] |
BARNARD A M L, BOWDEN S D, BURR T, et al, 2007. Quorum sensing, virulence and secondary metabolite production in plant soft-rotting bacteria[J]. Philosophical Transactions of the Royal Society B: Biological Sciences, 362(1483): 1165-1183.
doi: 10.1098/rstb.2007.2042 |
| [5] |
BASSLER B L, WRIGHT M, SHOWALTER R E, et al, 1993. Intercellular signalling in Vibrio harveyi: sequence and function of genes regulating expression of luminescence[J]. Molecular Microbiology, 9(4): 773-786.
doi: 10.1111/mmi.1993.9.issue-4 |
| [6] | CAO HUI, KRISHNAN G, GOUMNEROV B, et al, 2001. A quorum sensing-associated virulence gene of Pseudomonas aeruginosa encodes a LysR-like transcription regulator with a unique self-regulatory mechanism[J]. Proceedings of the National Academy of Sciences of the United States of America, 98(25): 14613-14618. |
| [7] |
CHEN XIN, SCHAUDER S, POTIER N, et al, 2002. Structural identification of a bacterial quorum-sensing signal containing boron[J]. Nature, 415(6871): 545-549.
doi: 10.1038/415545a |
| [8] |
CORNFORTH D M, FOSTER K R, 2013. Competition sensing: the social side of bacterial stress responses[J]. Nature Reviews Microbiology, 11(4): 285-293.
doi: 10.1038/nrmicro2977 |
| [9] |
DE KIEVIT T R, IGLEWSKI B H, 2000. Bacterial quorum sensing in pathogenic relationships[J]. Infection and Immunity, 68(9): 4839-4849.
doi: 10.1128/IAI.68.9.4839-4849.2000 |
| [10] |
DEFOIRDT T, 2018. Quorum-sensing systems as targets for antivirulence therapy[J]. Trends in Microbiology, 26(4): 313-328.
doi: 10.1016/j.tim.2017.10.005 |
| [11] |
EBERHARD A, BURLINGAME A L, EBERHARD C, et al, 1981. Structural identification of autoinducer of Photobacterium fischeri luciferase[J]. Biochemistry, 20(9): 2444-2449.
doi: 10.1021/bi00512a013 |
| [12] |
GROISMAN E A, 2016. Feedback control of two-component regulatory systems[J]. Annual Review of Microbiology, 70: 103-124.
doi: 10.1146/micro.2016.70.issue-1 |
| [13] |
GUPTA A, REIZMAN I M B, REISCH C R, et al, 2017. Dynamic regulation of metabolic flux in engineered bacteria using a pathway-independent quorum-sensing circuit[J]. Nature Biotechnology, 35(3): 273-279.
doi: 10.1038/nbt.3796 |
| [14] |
HANCOCK R E W, 2014. Collateral damage[J]. Nature Biotechnology, 32(1): 66-68.
doi: 10.1038/nbt.2779 |
| [15] |
HIBBING M E, FUQUA C, PARSEK M R, et al, 2010. Bacterial competition: surviving and thriving in the microbial jungle[J]. Nature Reviews Microbiology, 8(1): 15-25.
doi: 10.1038/nrmicro2259 |
| [16] |
HIGGINS D A, POMIANEK M E, KRAML C M, et al, 2007. The major Vibrio cholerae autoinducer and its role in virulence factor production[J]. Nature, 450(7171): 883-886.
doi: 10.1038/nature06284 |
| [17] |
KELLY R C, BOLITHO M E, HIGGINS D A, et al, 2009. The Vibrio cholerae quorum-sensing autoinducer CAI-1: analysis of the biosynthetic enzyme CqsA[J]. Nature Chemical Biology, 5(12): 891-895.
doi: 10.1038/nchembio.237 |
| [18] |
KUMAR S, STECHER G, TAMURA K, 2016. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets[J]. Molecular Biology and Evolution, 33(7): 1870-1874.
doi: 10.1093/molbev/msw054 |
| [19] |
LEE J, WU JIEN, DENG YINYUE, et al, 2013. A cell-cell communication signal integrates quorum sensing and stress response[J]. Nature Chemical Biology, 9(5): 339-343.
doi: 10.1038/nchembio.1225 |
| [20] | LETUNIC I, BORK P, 2018. 20 years of the SMART protein domain annotation resource[J]. Nucleic Acids Research, 46(D1): D493-D496. |
| [21] |
MILLER M B, BASSLER B L, 2001. Quorum sensing in bacteria[J]. Annual Review of Microbiology, 55: 165-199.
doi: 10.1146/micro.2001.55.issue-1 |
| [22] |
MILLER M B, SKORUPSKI K, LENZ D H, et al, 2002. Parallel quorum sensing systems converge to regulate virulence in Vibrio cholerae[J]. Cell, 110(3): 303-314.
doi: 10.1016/S0092-8674(02)00829-2 |
| [23] |
MITRA A, HERREN C D, PATEL I R, et al, 2016. Integration of AI-2 based cell-cell signaling with metabolic cues in Escherichia coli[J]. PLoS One, 11(6): e0157532.
doi: 10.1371/journal.pone.0157532 |
| [24] | MONNET V, JUILLARD V, GARDAN R, 2016. Peptide conversations in Gram-positive bacteria[J]. Critical Reviews in Microbiology, 42(3): 339-351. |
| [25] | MOREIRA C G, SPERANDIO V, 2016. The Epinephrine/ Norepinephrine/Autoinducer-3 interkingdom signaling system in Escherichia coli O157:H7[M]//LYTE M. Microbial endocrinology:interkingdom signaling in infectious disease and health. Cham: Springer, 874: 247-261. |
| [26] |
MUKHERJEE S, BASSLER B L, 2019. Bacterial quorum sensing in complex and dynamically changing environments[J]. Nature Reviews Microbiology, 17(6): 371-382.
doi: 10.1038/s41579-019-0186-5 |
| [27] |
NA S I, KIM Y O, YOON S H, et al, 2018. UBCG: Up-to-date bacterial core gene set and pipeline for phylogenomic tree reconstruction[J]. Journal of Microbiology, 56(4): 280-285.
doi: 10.1007/s12275-018-8014-6 |
| [28] |
NG W L, BASSLER B L, 2009. Bacterial quorum-sensing network architectures[J]. Annual Review of Genetics, 43: 197-222.
doi: 10.1146/genet.2009.43.issue-1 |
| [29] |
NG W L, PEREZ L J, WEI YUNZHOU, et al, 2011. Signal production and detection specificity in Vibrio CqsA/CqsS quorum-sensing systems[J]. Molecular Microbiology, 79(6): 1407-1417.
doi: 10.1111/mmi.2011.79.issue-6 |
| [30] |
NOVAK E A, SHAO HANJUAN, DAEP C A, et al, 2010. Autoinducer-2 and QseC control biofilm formation and in vivo virulence of Aggregatibacter actinomycetemcomitans[J]. Infection and Immunity, 78(7): 2919-2926.
doi: 10.1128/IAI.01376-09 |
| [31] |
PAPENFORT K, BASSLER B L, 2016. Quorum sensing signal-response systems in Gram-negative bacteria[J]. Nature Reviews Microbiology, 14(9): 576-588.
doi: 10.1038/nrmicro.2016.89 |
| [32] |
PEREIRA C S, DE REGT A K, BRITO P H, et al, 2009. Identification of functional LsrB-like autoinducer-2 receptors[J]. Journal of Bacteriology, 191(22): 6975-6987.
doi: 10.1128/JB.00976-09 |
| [33] |
PEREIRA C S, THOMPSON J A, XAVIER K B, 2013. AI-2-mediated signalling in bacteria[J]. FEMS Microbiology Reviews, 37(2): 156-181.
doi: 10.1111/j.1574-6976.2012.00345.x |
| [34] | PESCI E C, MILBANK J B J, PEARSON J P, et al, 1999. Quinolone signaling in the cell-to-cell communication system of Pseudomonas aeruginosa[J]. Proceedings of the National Academy of Sciences of the United States of America, 96(20): 11229-11234. |
| [35] |
RAINEY P B, RAINEY K, 2003. Evolution of cooperation and conflict in experimental bacterial populations[J]. Nature, 425(6953): 72-74.
doi: 10.1038/nature01906 |
| [36] |
RAJEEV L, GARBER M E, MUKHOPADHYAY A, 2020. Tools to map target genes of bacterial two-component system response regulators[J]. Environmental Microbiology Reports, 12(3): 267-276.
doi: 10.1111/emi4.v12.3 |
| [37] |
SCHÄFFER A A, ARAVIND L, MADDEN T L, et al, 2001. Improving the accuracy of PSI-BLAST protein database searches with composition-based statistics and other refinements[J]. Nucleic Acids Research, 29(14): 2994-3005.
doi: 10.1093/nar/29.14.2994 |
| [38] | SCHOLZ R L, GREENBERG E P, 2017. Positive autoregulation of an acyl-homoserine lactone quorum-sensing circuit synchronizes the population response[J]. mBio, 25; 8(4): e01079-17. |
| [39] | SPERANDIO V, TORRES A G, JARVIS B, et al, 2003. Bacteria-host communication: the language of hormones[J]. Proceedings of the National Academy of Sciences of the United States of America, 100(15): 8951-8956. |
| [40] |
STEPHENS K, BENTLEY W E, 2020. Synthetic biology for manipulating quorum sensing in microbial consortia[J]. Trends in Microbiology, 28(8): 633-643.
doi: 10.1016/j.tim.2020.03.009 |
| [41] |
TAGA M E, MILLER S T, BASSLER B L, 2003. Lsr-mediated transport and processing of AI-2 in Salmonella typhimurium[J]. Molecular Microbiology, 50(4): 1411-1427.
doi: 10.1046/j.1365-2958.2003.03781.x |
| [42] |
TANG J L, LIU Y N, BARBER C E, et al, 1991. Genetic and molecular analysis of a cluster of rpf genes involved in positive regulation of synthesis of extracellular enzymes and polysaccharide in Xanthomonas campestris pathovar campestris[J]. Molecular and General Genetics MGG, 226(3): 409-417.
doi: 10.1007/BF00260653 |
| [43] |
URBANCZYK H, AST J C, HIGGINS M J, et al, 2007. Reclassification of Vibrio fischeri, Vibrio logei, Vibrio salmonicida and Vibrio wodanis as Aliivibrio fischeri gen. nov., comb. nov., Aliivibrio logei comb. nov., Aliivibrio salmonicida comb. nov. and Aliivibrio wodanis comb. nov[J]. International Journal of Systematic and Evolutionary Microbiology, 57(12): 2823-2829.
doi: 10.1099/ijs.0.65081-0 |
| [44] |
VON BODMAN S B, WILLEY J M, DIGGLE S P, 2008. Cell-cell communication in bacteria: united we stand[J]. Journal of Bacteriology, 190(13): 4377-4391.
doi: 10.1128/JB.00486-08 |
| [45] |
WANG FANGFANG, QIAN WEI, 2019. The roles of histidine kinases in sensing host plant and cell-cell communication signal in a phytopathogenic bacterium[J]. Philosophical Transactions of the Royal Society B: Biological Sciences, 374(1767): 20180311.
doi: 10.1098/rstb.2018.0311 |
| [46] | WANG YANG, LIU BAOBAO, GRENIER D, et al, 2019. Regulatory mechanisms of the LuxS/AI-2 system and bacterial resistance[J]. Antimicrobial Agents and Chemotherapy, 63(10): e01186-19. |
| [47] |
WEI YUNZHOU, NG W L, CONG JIANPING, et al, 2012. Ligand and antagonist driven regulation of the Vibrio cholerae quorum-sensing receptor CqsS[J]. Molecular Microbiology, 83(6): 1095-1108.
doi: 10.1111/mmi.2012.83.issue-6 |
| [48] |
WEST S A, GRIFFIN A S, GARDNER A, et al, 2006. Social evolution theory for microorganisms[J]. Nature Reviews Microbiology, 4(8): 597-607.
doi: 10.1038/nrmicro1461 |
| [49] |
WHITELEY M, DIGGLE S P, GREENBERG E P, 2017. Progress in and promise of bacterial quorum sensing research[J]. Nature, 551(7680): 313-320.
doi: 10.1038/nature24624 |
| [50] |
WINZER K, HARDIE K R, BURGESS N, et al, 2002a. LuxS: its role in central metabolism and the in vitro synthesis of 4-hydroxy-5-methyl-3(2H)-furanone[J]. Microbiology, 148(4): 909-922.
doi: 10.1099/00221287-148-4-909 |
| [51] |
WINZER K, HARDIE K R, WILLIAMS P. 2002b. Bacterial cell-to-cell communication: sorry, can't talk now - gone to lunch![J]. Current Opinion in Microbiology, 5(2): 216-222.
doi: 10.1016/S1369-5274(02)00304-1 |
| [52] |
WU SHENGBO, LIU JIAHENG, LIU CHUNJIANG, et al, 2020. Quorum sensing for population-level control of bacteria and potential therapeutic applications[J]. Cellular and Molecular Life Sciences, 77(7): 1319-1343.
doi: 10.1007/s00018-019-03326-8 |
| [53] |
ZIMMERMANN L, STEPHENS A, NAM S Z, et al, 2018. A completely reimplemented MPI bioinformatics toolkit with a new HHpred server at its core[J]. Journal of Molecular Biology, 430(15): 2237-2243.
doi: 10.1016/j.jmb.2017.12.007 |
| [1] | 穆蓉, 朱珠, 张瑞峰. 基于比较基因组学分析海洋聚球藻对铁限制的适应机制[J]. 热带海洋学报, 2023, 42(6): 89-100. |
| [2] | 张婷, 王瑞旋, 孙敬锋, 黄嘉健, 林小植, 周飞, 马细兰. 哈维弧菌对皱纹盘鲍不同组织内细菌群落结构的影响[J]. 热带海洋学报, 2023, 42(1): 56-65. |
| [3] | 缪莉, 钱嘉兴, 莫杰, 周恒, 钱生辉, 董昆明. 海洋放线菌Nocardiopsis dassonvillei JS106的抗群体感应活性物质研究[J]. 热带海洋学报, 2023, 42(1): 145-151. |
| [4] | 喻飞, 金兴坤, 雷天影, 曹海航, 陈锵辉, 阳耀帆, 李佳航, 赵哲. 海洋弧菌HN897 β-琼脂糖酶基因vas1-1339异源表达及活性分析[J]. 热带海洋学报, 2022, 41(2): 170-176. |
| [5] | 朱馨媛, 刘敏, 黄颖, 赵哲. 含铁肠杆菌素受体调节蛋白VPA0148对副溶血弧菌毒力的影响[J]. 热带海洋学报, 2021, 40(6): 93-101. |
| [6] | 李炳, 王瑞旋, 张立, 罗帮, 牟红莉, 王江勇. 北部湾养殖牡蛎体内异养细菌数量及其耐药性研究[J]. 热带海洋学报, 2021, 40(4): 70-83. |
| [7] | 钟宛宣, 杨芸兰, 李祥付, 徐杰. 两株溶藻弧菌噬菌体的生理特性和基因组研究[J]. 热带海洋学报, 2021, 40(4): 22-34. |
| [8] | 李艳群, 陈柔雯, 林宗豪, 田新朋, 尹浩. 一株群体感应抑制活性海洋放线菌的筛选与鉴定 *[J]. 热带海洋学报, 2021, 40(1): 75-81. |
| [9] | 季宇彬,张哲,张伟浩,王虎,潘英,姜薇. 群体感应抑制活性导向分离腐皮镰刀菌中代谢产物[J]. 热带海洋学报, 2019, 38(3): 98-103. |
| [10] | 陆友云, 薛明, 李志桦, 温崇庆. 海洋蛭弧菌DA5全基因组测序及序列分析[J]. 热带海洋学报, 2018, 37(6): 112-119. |
| [11] | 陈政强,陈昌生,战文斌,林茂,杨红玲,. 饥饿胁迫对九孔鲍免疫防御因子的影响*[J]. 热带海洋学报, 2012, 31(5): 124-130. |
| [12] | 李海平 ,鄢庆枇 ,徐晓津 ,苏永全 ,覃映雪 . 哈维氏弧菌生物被膜体外模型的建立及成膜特性研究[J]. 热带海洋学报, 2011, 30(3): 99-104. |
| [13] | 温崇庆,梁华芳,丁贤,薛明,周世宁. 海洋蛭弧菌类生物DA5对凡纳滨对虾育苗期幼体和水质的影响[J]. 热带海洋学报, 2010, 29(6): 147-152. |
| [14] | 梅冰,周永灿,徐先栋,王世峰,谢珍玉. 斜带石斑鱼烂尾病病原菌的分离与鉴定[J]. 热带海洋学报, 2010, 29(6): 118-124. |
| [15] | 杜虹,黄显兵,郑兵,陈伟洲,庄东红,胡忠. 粤东深澳湾养殖区域异养细菌和弧菌的动态分布[J]. 热带海洋学报, 2010, 29(6): 110-117. |
|
||